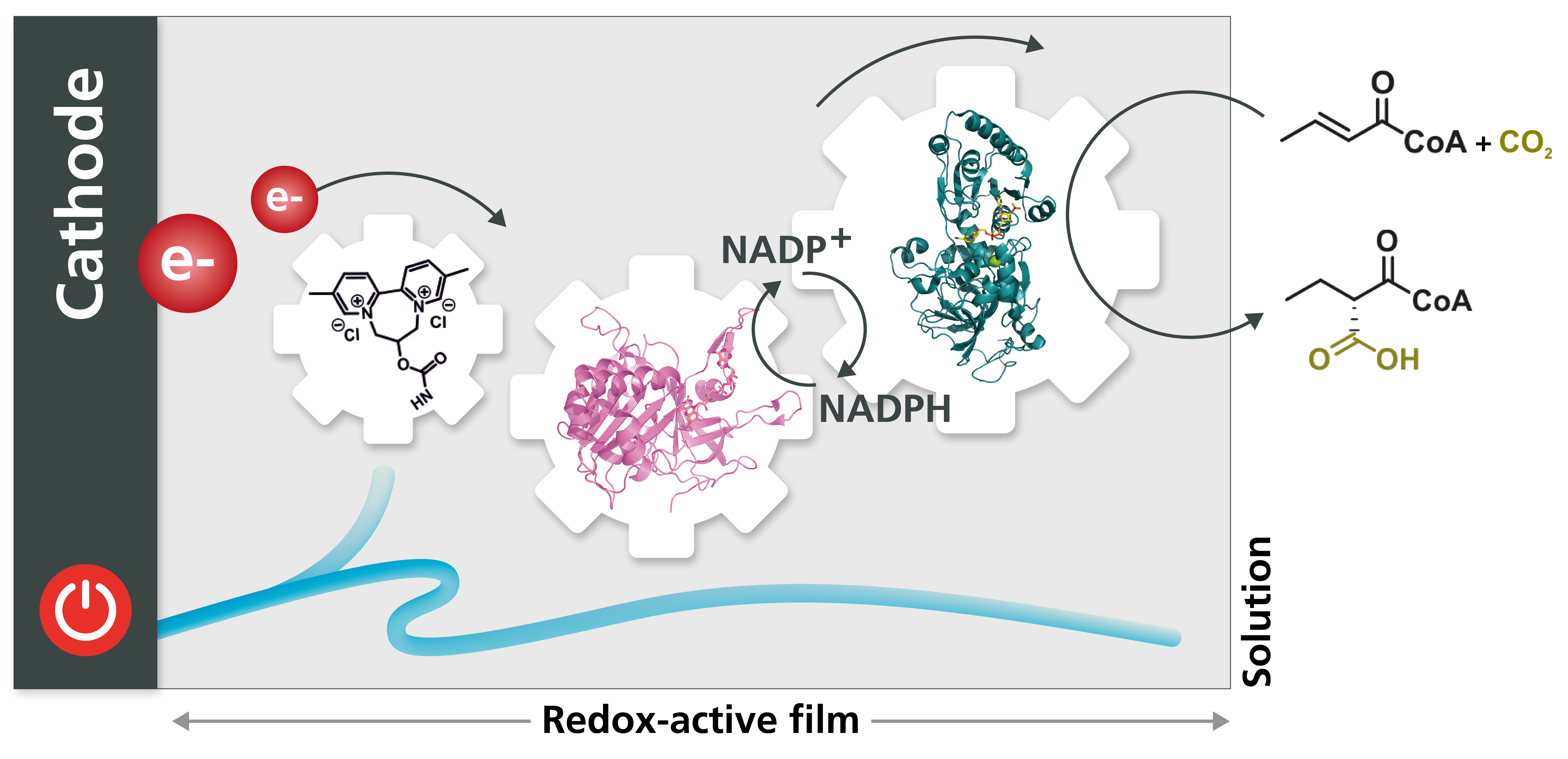
Regenerating the cofactor
For the reductive carboxylation reaction to take place, the enzyme enoyl-CoA-carboxylase/reductase requires NADPH as a cofactor. This small, organic molecule acts as either a proton or electron donor, and is used up in the course of every individual reaction. Providing new cofactors every time the synthetic reaction is conducted would be very expensive on a large scale, and as such, it would not be financially viable. As a result, the project team had to find a way of using electricity and the same reaction principles to regenerate the cofactor molecules, so that they would be available for further reaction cycles. Ultimately, they hit on the solution of immobilizing another enzyme in the redox hydrogel to act as a recycling module.
Demonstrating CO2 incorporation into crotonyl-CoA
With this new approach combining CO2 reduction, bioelectrocatalytic technology, material chemistry and synthetic biology, the scientists were able to demonstrate the feasibility of the process at a milliliter scale. By enabling regioselective and stereoselective incorporation of CO2 into crotonyl-CoA, the new approach delivered the most complex product ever achieved in the biocatalytic conversion of CO2. The results of the studies were published in 2021 in “Angewandte Chemie,” a scientific journal for applied chemistry, and selected as a “hot paper.”
Modular platform technology
As the electro-biocatalytic reaction system can be extended on a modular basis, it can function as a platform technology. Depending on the target molecule (synthesis product), suitable enzymes can be selected from bioinformatic databases, manufactured using biotechnology, and embedded in the hydrogels. Consequently, the eBioCO2n module can enable the manufacture of various biobased fine chemicals that can be diversified as needed using specific enzyme cascades. This holds great application potential by companies in the pharmaceutical, agrochemical and food industries.
Outlook
In the next steps, the team aims to complete the scaling and modular expansion of the process. Chemical synthesis based on biocatalytic CO2 fixation is paving the way for a new form of circular economy by offering alternatives to fossil-based raw materials in chemical synthesis processes.
 Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB
Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB