Membranes for the hydrogen economy
Membranes are expected to play an important role at many points in the production and use of green hydrogen. This starts with the provision of clean water for electrolysis (Water purification), to the electrochemical splitting of water in polymer electrolyte membrane (PEM) electrolyzers, to the reconversion to electricity using PEM fuel cells.
Membrane humidifiers, which we are developing at the IGB and testing in automated test rigs (Moisture management), can also be used for water management in fuel cells.
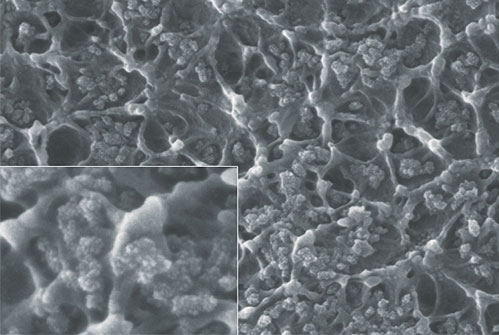
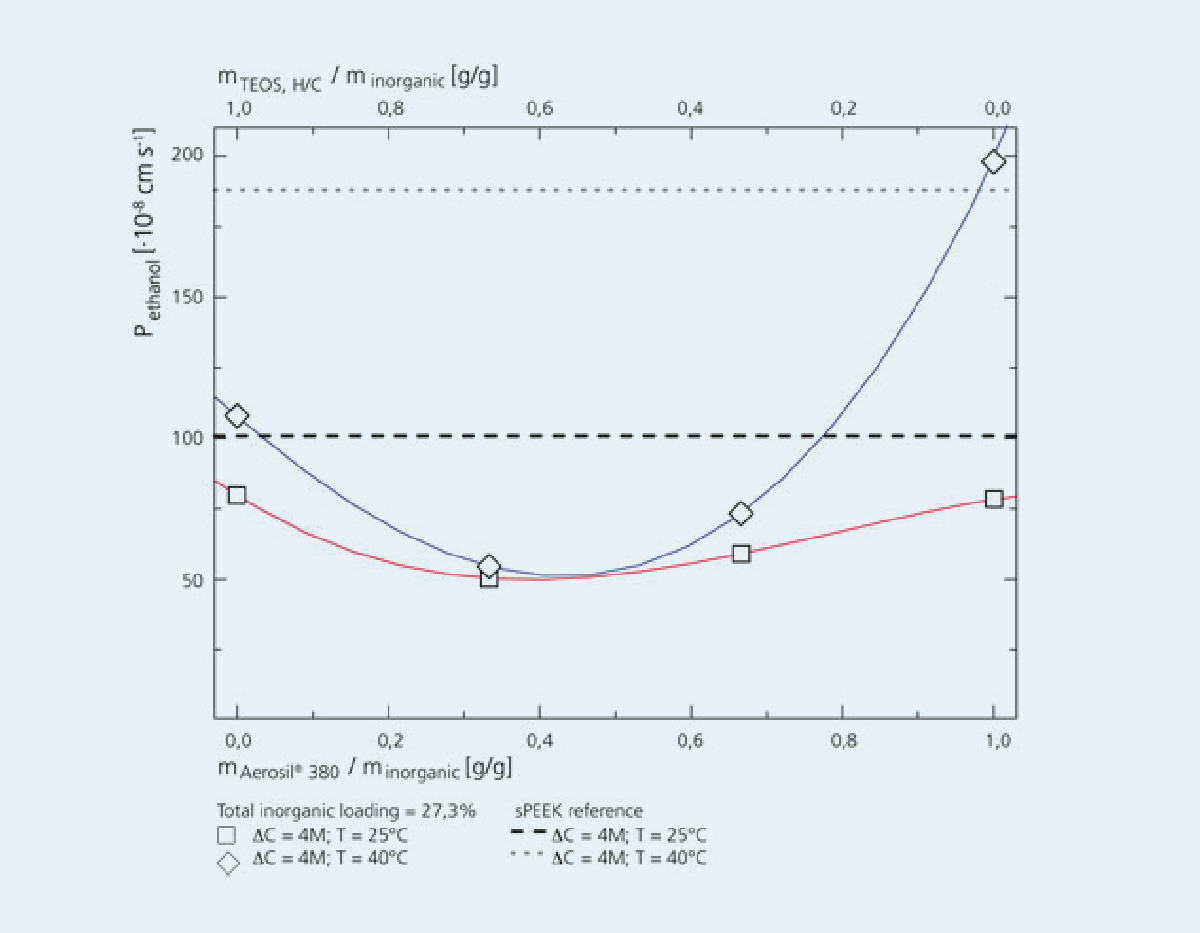
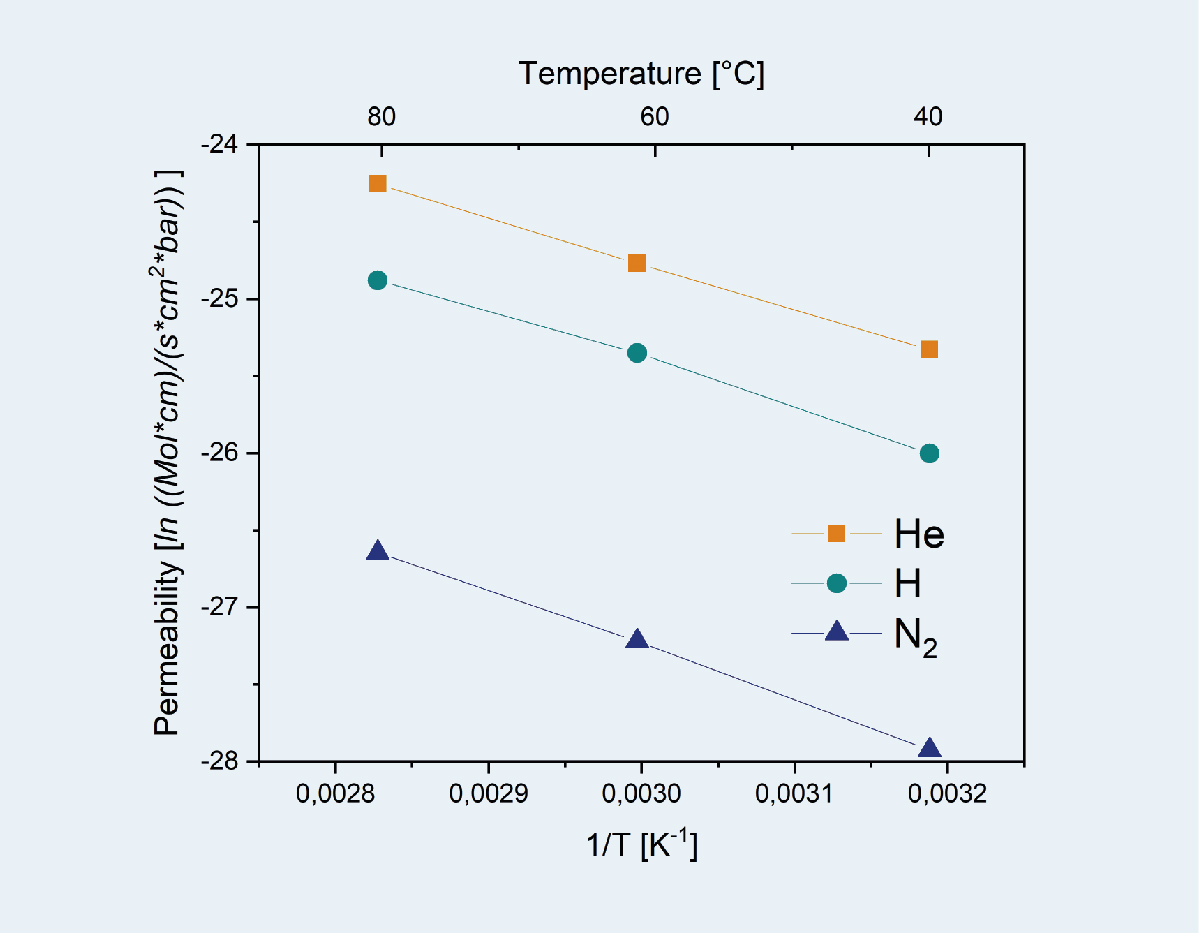
Other possible applications for membranes are the separation of H2-containing gas mixtures by means of membranes, such as palladium membranes (Gas separation) or the production of hydrogen in so-called membrane reactors by direct splitting of water (Oxygen conducting perovskite capillary membranes).
 Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB
Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB