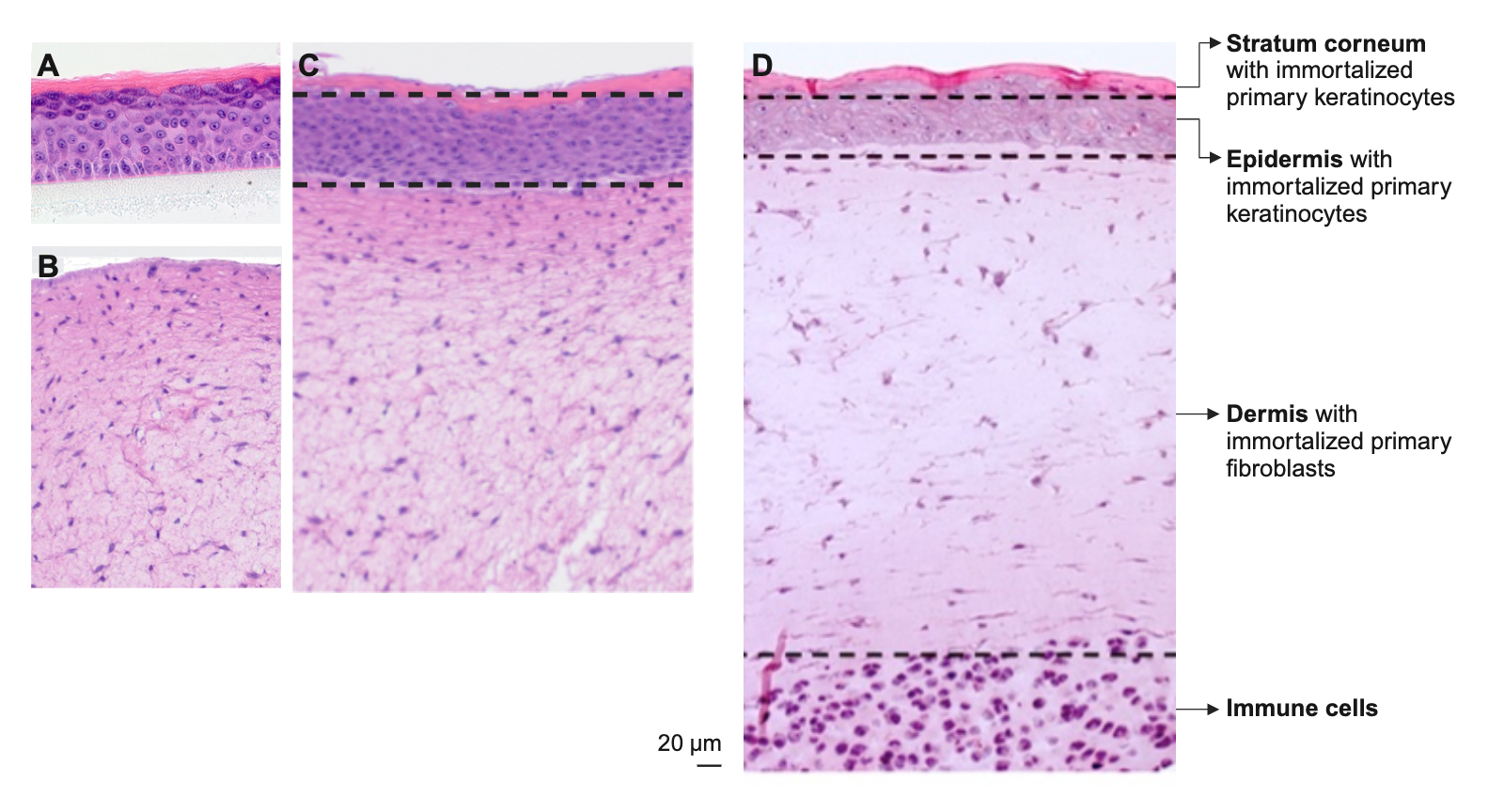
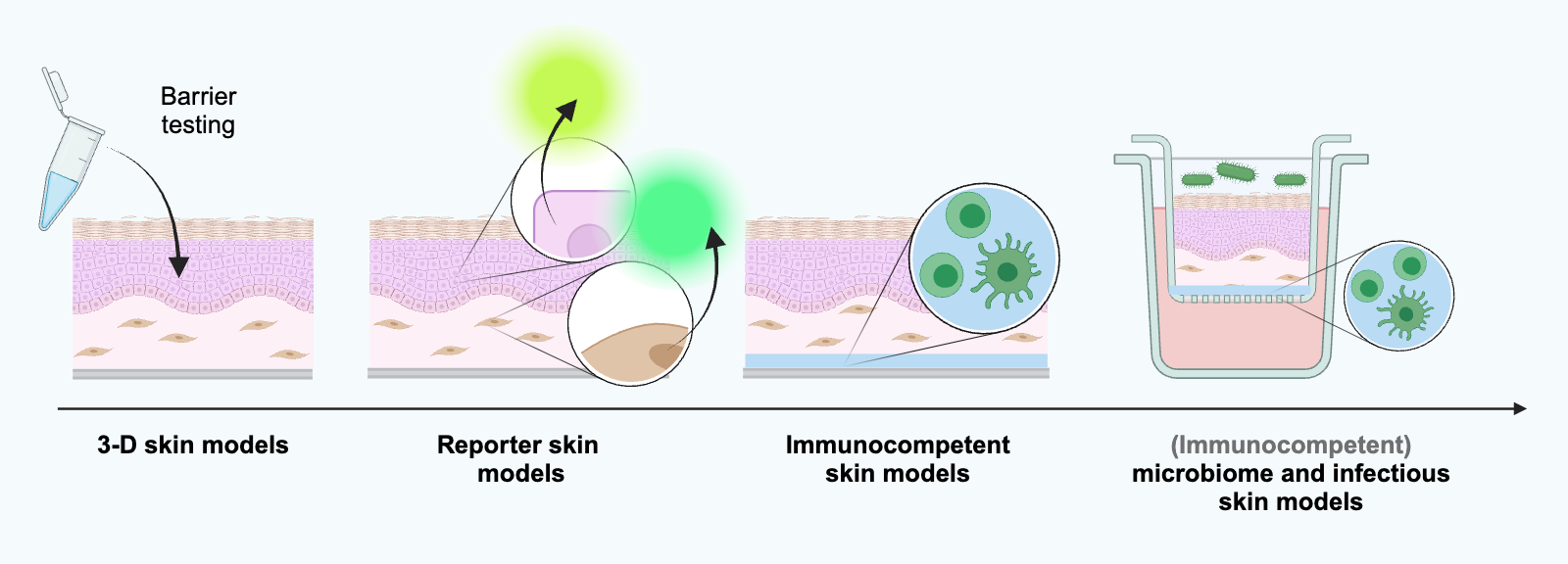
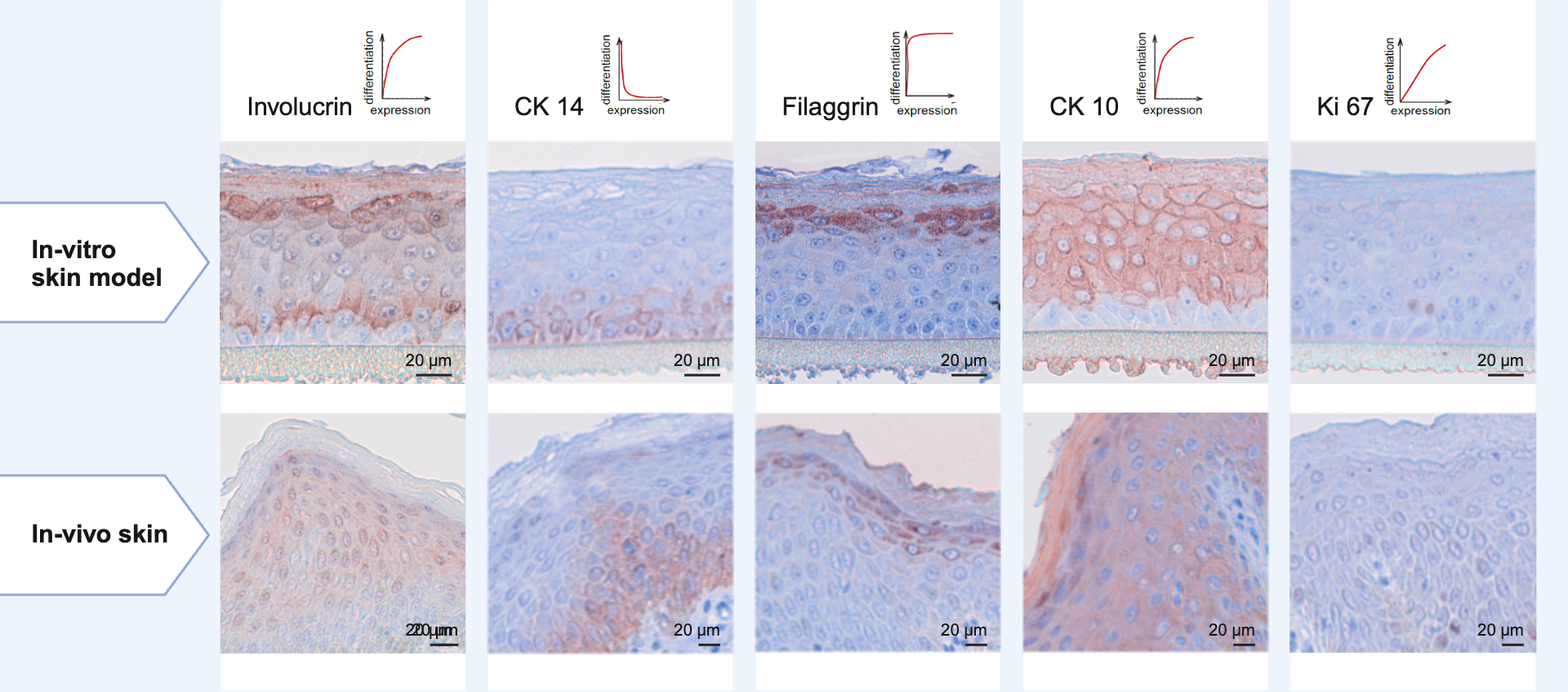
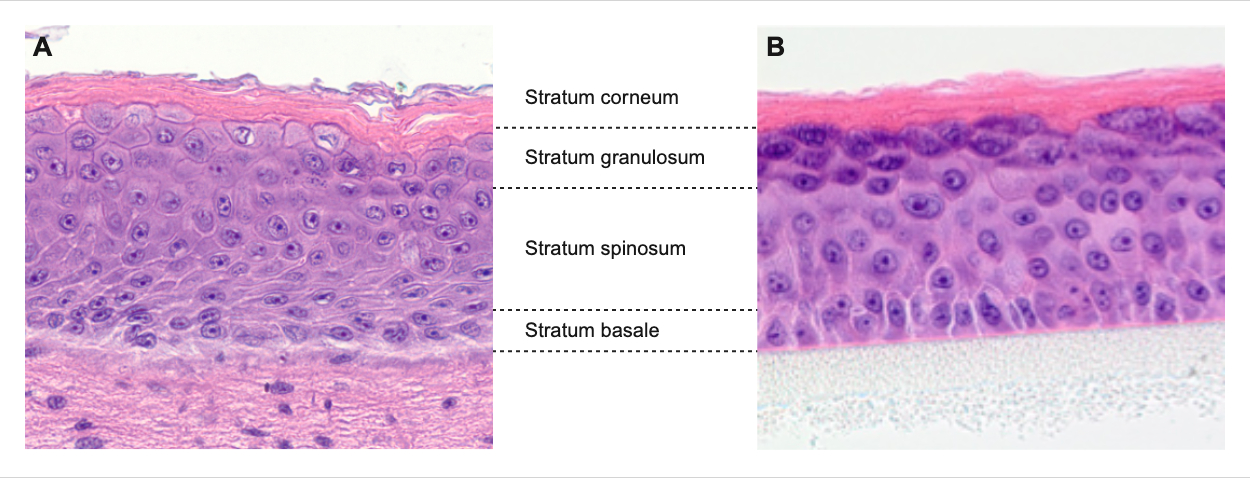
We develop metabolically active 3D skin models of varying complexity as in vitro test systems based on immortalized primary or primary cells:
Reconstituted dermis models,
which are constructed from immortalized primary or primary fibroblasts in a collagen layer on a porous polycarbonate membrane and are cultivated submerged.
Reconstituted epidermis models,
which are constructed from immortalized primary or primary keratinocytes on a porous polycarbonate membrane and cultured at the air-liquid interface.
Reconstituted full-thickness skin models,
composed of immortalized primary or primary fibroblasts and immortalized primary or primary keratinocytes. The dermal tissue equivalent of fibroblasts is cultured on a porous polycarbonate membrane. This is covered by an epidermal layer of differentiated immortalized primary or primary keratinocytes. The model is cultivated at the air-liquid interface.
Immunocompetent full-thickness skin models,
which are constructed from reconstituted full-thickness skin models, supplemented with immune cells such as peripheral blood mononuclear cells (PBMCs) and cultivated at the air-liquid interface on a porous polycarbonate membrane.
 Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB
Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB