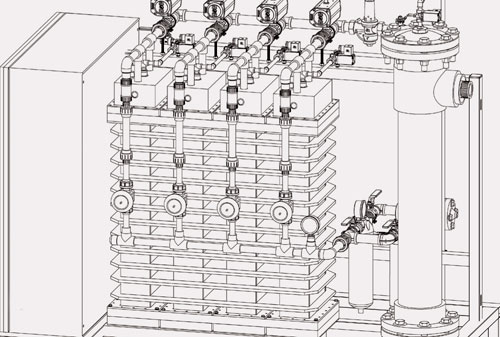
The removal of pollutants in low concentrations, known as organic trace substances or micropollutants, is becoming an increasingly important priority in water treatment.
Removal of micropollutants in wastewater and process water with AOP
Oxidative water treatment (AOP, advanced oxidation processes) is understood as processes for chemical water treatment in which hydroxyl radicals are formed. These highly reactive radicals are available for chemical decomposition reactions and react with organic or inorganic substances that are not easy to break down biologically. They can be formed by adding oxidative substances such as ozone and hydrogen peroxide, or by introducing energy by means of UV radiation, ultrasound or electric current via inert electrodes as well as by a combination of these processes.
At present, catalytic, photochemical, and electrochemical processes as well as plasma processes for oxidative water treatment are investigated at Fraunhofer IGB. Various experimental set-ups for continuous, semi-continuous and batch trials are available for this purpose.
Advantages and application areas
AOP processes are always used when a biological decomposition is not feasible or cannot be carried out efficiently, for example because the contaminations contain persistent substances. Also, AOP processes are the method of choice when the process wastewater has a toxic effect on the microorganisms of a biological purification stage or occurs extremely discontinuously. The energy required for operating the system can be provided by electricity from renewable sources such as solar and wind power.
 Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB
Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB