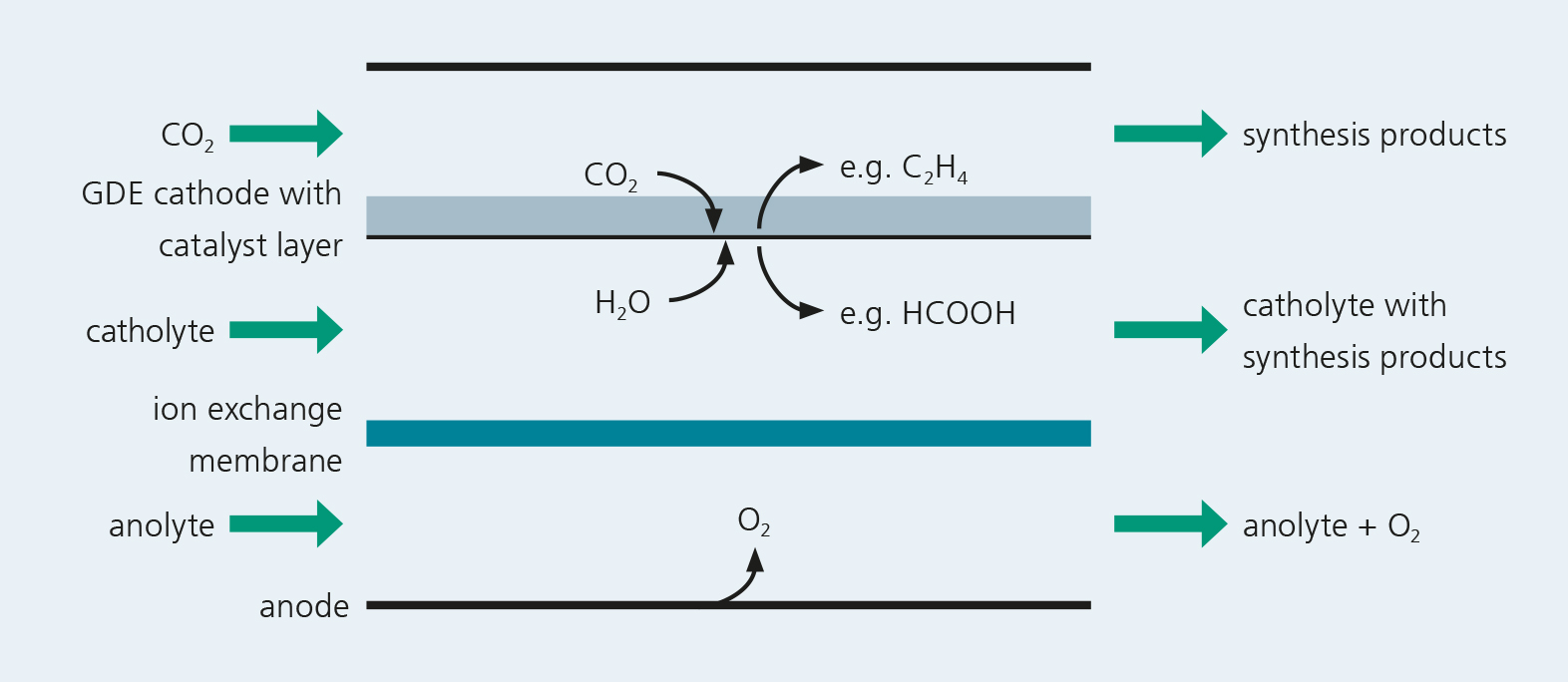
Challenges
Utilization of electric energy for the production of basic chemicals is gaining increasing economic relevance. The electrosynthesis of essential chemical products can solve two current challenges:
Chemical storage of volatile renewable energy
First, the ongoing energy transition leads to a massive expansion of renewable energy and therefore to an increasing but fluctuating supply of electric energy, which requires new storage and utilization concepts. In periods of low demand for electricity, the excess supply of electrical energy can be alternatively used for the production of value.
Chemicals from electricity replace fossil raw materials
Second, many industrial processes depend on basic chemicals that are currently produced from fossil and mainly imported resources, entailing substantial costs and effort for purchasing, transport and storage.
 Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB
Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB